Transcription
Malays. Appl. Biol. (2016) 45(1): 69–80ACTIVE COMPOUND, ANTIOXIDANT, ANTIPROLIFERATIVEAND EFFECT ON STZ INDUCED ZEBRAFISH OF VARIOUSCRUDE EXTRACTS FROM Boletus qriseipurpureusSUTHA MUNIANDY1, FAUZI DAUD1, SAHIDAN SENAFI1, MAHANEM MAT NOOR1,MALINA KUMARAN1, ANDI NORAFIQAH ANDIK MOHD ALWI1,AYUMAWARNI YUSNI YENG JEE LONG1, RAHIMAH BINTI MOHAMAD ZUKI1,JANET SALOMI ISAC1, SUBHASHINI KRISHNAN1, DEEVIYA GOBALAN1, YI CHIEH LIM1,AZWAN MAT LAZIM2 and SHAZRUL FAZRY1*1Schoolof Bioscience and Biotechnology, Faculty of Science and Technology,Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia.2School of Chemical Sciences and Food Technology, Faculty of Science and Technology,Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia*Email: shazrul@ukm.edu.myABSTRACTBoletus qriseipurpureus (gelam mushroom) is a mushroom used by locals in Tok Bali, Kelantan, Malaysia to treat diabetes,cervical cancer and breast cancer. The active compounds in B. qriseipurpureus remain unidentified. Therefore, in this study,we investigated the potential medicinal properties of B. qriseipurpureus extracts (hot water, cold water and methanol extracts)by conducting preliminary phytochemical screening, in vitro antioxidant, in vitro antiproliferative and in vivo antidiabetic test.Biochemical assays were performed to detect the presence of alkaloids, anthraquinones, flavonoids, reducing sugars, saponins,steroids and tannins in B. qriseipurpureus extracts. 1,1-diphenyl-2-picrylhydrazyl (DPPH) was used to evaluate the antioxidantcapacity of B. qriseipurpureus extracts, while MTT cell proliferation assay umbromide) was used to investigate the extracts antiproliferative activity. The effects of B. qriseipurpureus extracts onstreptozotocin (STZ) induced zebrafish were examined at the concentration of 45 mg/mL, 90 mg/mL and 135 mg/mL. Theeffect of extracts were measured by the regenerative growth rate of the amputated caudal fin for fourteen days post transection.Screening of the mushroom extracts for active compounds revealed the presence of alkaloids, flavonoids, saponins and tanninsin all test extracts. Reducing sugars and anthraquinones are only detected in hot water and cold water of B. qriseipurpureusextracts. The half maximal inhibitory concentration (IC50) of DPPH by hot water, cold water and methanol extract of B.qriseipurpureus are 1.79 mg/mL, 1.97 mg/mL and 3.98 mg/mL respectively. The MTT assay indicated that all extracts exhibitedsignificant antiproliferative effects on MCF-7 cell line after 72 hours with IC50 of 7.7 mg/mL for hot water extract, 8.2 mg/mL for cold water extract and 16.1 mg/mL for methanol extract but do not display any significant cytotoxic effect. The STZinduced zebrafish treated with 135 mg/mL of hot water B. qriseipurpureus extract for 14 days showed the highest regenerationgrowth rate of caudal fin (5.04 0.43%) compared to fish treated with metformin (5.72 0.64 %). In this study, we showedthe potential of hot water B. qriseipurpureus extracts as a potent therapeutic agent for diabetes and as an alternative naturalsource of antioxidant.Key words: Boletus qriseipurpureus, active compound, antioxidant, antiproliferative, antidiabetic, zebrafishINTRODUCTIONIn recent years, mushrooms have attracted researchesattentions for their functional properties in medicalapplication (Sun et al., 2011). Some mushroomshave functional roles in antinflammatory, antitumor,anticancer, antibacterial, antioxidant, antiviral,antiallergic, hypoglycemic and haematological* To whom correspondence should be addressed.properties (Barros et al., 2007a; Barros et al., 2007b;Chen et al., 2009; Dore et al., 2007; Faccin et al.,2007; Garcia et al., 2010; Guillamon et al., 2010;Yoshino et al., 2008).B. qriseipurpureus is a species of wild ediblemushroom found along the coast from Bachok,Kelantan to Kuala Terengganu. This mushroom islocally known as “Cendawan (malay word formushroom) Gelam” as it grows in the sandy areawith gelam trees (Melaleuca cajuput Powell). It isused by the locals to treat diabetes, cervical cancer
70STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusand breast cancer. Methanol extract of B.qriseipurpureus was shown to be rich withnatural antioxidant metabolites such as 2,4,6trimethylacetophenone imine, glutamyl tryptophan,azatadine and lithocholic acid glycine conjugate(Yuswan et al., 2015). We recently conducted astudy on B. qriseipurpureus extracts (hot water, coldwater and methanol) to determine the effect ofextracts on diabetes related enzymes (alpha-amylaseand alpha-glucosidase) (Muniandy et al., 2015).The study shows that hot water extract possessedthe highest inhibition activity of α-amylase andα-glucosidase in a concentration dependentmanner with the IC 50 value 87 mg/mL and 89mg/mL respectively. The study also suggested thathot water extract of B. qriseipurpureus containsbioactive compound that can inhibit α-amylase andα-glucosidase enzyme activity.Since diabetes has been related to antioxidant,and antioxidant has been correlated withantiproliferation (Fatimah et al., 2012; Wing et al.,2007), here we determined the active compound,antioxidant, antiproliferative and antidiabeticproperties of B. qriseipurpureus extracts. Toinvestigate the antidiabetic properties of theseextracts in vivo, tail regeneration assay ofstreptozotocin (STZ), a diabetogenic drug whichdamage insulin-producing beta cells of the pancreas(Rossini et al., 1977) induced zebrafish was utilisedas described by Robert et al. (2013).MATERIALS AND METHODSSample Preparation and ExtractionWild B. qriseipurpureus were forage from therural area of Tok Bali, Bachok, Kelantan. Extractsof B. qriseipurpureus were prepared according toAhmad et al. (2014) with slight modification. Allmushrooms used in the extraction were freeze-driedimmediately after harvest. For the cold water (28 2ºC) and methanol extraction, 50 g of blendmushroom sample was soaked in 200 mL of coldwater and methanol, respectively. The mushroomswere left for 36 hours at room temperature (28 2ºC)with continuous shaking at 200 rpm. For hot waterextraction, a total of 50 g of powdered sample wasimmersed in 200 mL of hot water (95 5ºC) andleft for 4 hours at room temperature with continuousshaking at 200 rpm. The hot water and cold waterextracts were filtered using filter paper (WhatmanNo1). The filtrates were then freeze-dried undervacuum condition at -50ºC for five days. Themethanol was evaporated from the methanol extractusing rotary evaporation technique at 50ºC with aspeed of 90 rpm until completely dry. All extractswere stored at 4ºC in a clean sterile container forfurther use.Mycochemical ScreeningAlkaloidsAlkaloids detection was performed according tothe method described by Kasolo et al. (2010).Extract (0.01 g) was added to 3 drops of 2%hydrochloric acid. 3 drops Dragendorff reagent wasadded to the solution, precipitating the alkaloids.AnthraquinonesAnthraquinones detection was performedaccording to the method described by Ayoola et al.(2008). Extract (0.1 g) was added to 2 mL of 10%hydrochloric acid and heated for 2 minutes. Thesolution was filtered while warm. The liquid filtratewas then added with CH3Cl3 in a ratio of 1:1 andshaken. Then, a few drops of 10% ammonia wereadded. The presence of pink or red colour on thebottom of the test tube indicates the presence ofanthraquinones.FlavonoidsDetection of flavonoids was conducted usingShinoda test as described by Sabri et al. (2012).Extract (0.1 g) was added into a solution of 5 mL ofdistilled water and 1 ml of 10% H2SO4. About 0.5 gof magnesium was added into the solution. Thesolution turns red in presence of flavonoids.Reducing sugarsDetection of reducing sugars was carried outusing Fehling test as described by Sabri et al.(2012). Extract (0.1 g) was added into 1 mL ofdistilled water and 2 mL of mixed reagent FehlingA and B (what is the ratio of Fehling A to B/vol:vol). The mixture was then incubated at 60ºC. Brickred precipitation formed at the bottom of the testtube indicates the presence of reducing sugars.SaponinsSaponins detection was done according tothe method described by Adebayo et al. (2012).Extract (0.1 g) was mixed in 5 mL of water, andboiled for 2 minutes. Undissolved extract wereleft to precipitate at the bottom of the tube. Thesupernatant was used for the next step of the test.For the foaming test, 1 mL of the supernatant wasmixed with 4 mL of distilled water and shaken. Theformation of bubbles which last for 15 minutesindicates the presence of saponins. For the emulsiontest, 2 drops of olive oil was added to the liquidsolution of foaming test and mixed. The emulsionformation indicates the presence of saponins.SteroidsSteroids detection was conducted usingSalkowski test as described by Sharma et al. (2013).Extract (0.05 g) was added to 0.5 mL of distilled
STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureuswater. 2 mL of chloroform was then added to themixture, and mix by swirling the tube. 2 ml of 10%H2SO4 was added slowly to the mixture. Formationof red colour in the chloroform layer and the layerof yellowish green colour indicates the presence ofsteroids.TanninsTannins detection was performed using ferricchloride test as described by Ayoola et al. (2008).Extract (0.05 g) was added to 2 mL of distilledwater, boiled, and then filtered. Four drops of 0.1%FeCl3 was then added to the filtrate. The change ofcolour to blue-black, blue-green or green indicatesthe presence of tannins.Antioxidant Assay with DPPHDPPH assay was performed according to themethod proposed by Ahmad et al. (2014). Differentconcentrations of the crude extract (1 mL) indistilled water were added to 4 mL of 0.004% DPPHin methanol solution. The mixtures were incubatedat room temperature, in the dark for 30 minutesbefore reading the absorbance against a blank at517 nm. Inhibition of free radical by DPPH inpercent (I%) was calculated using the followingway:Scavenging effect (%) (ABlank – ASample / ABlank) 100Where ABlank is the absorbance of the controlreaction (containing all reagents except the testcompound), and ASample is the absorbance of the testcompound. The value of 50% inhibition (IC50) wascalculated from the graph plotted inhibitionpercentage against extract concentrations. Tests werecarried out in triplicate.Antiproliferative AssayAntiproliferative assay was conducted asdescribed by Shao et al. (2013) with slightmodification to determine the antiproliferativeeffects of B. qriseipurpureus extracts on Africangreen monkey’s kidney epithelial cell line (Vero)and breast cancer cell lines (MCF-7) using the MTTassay.All cells were grown in RPMI 1640 medium andmaintained in 37 C incubator with 5% CO 2. Tomeasure cell viability, 1 105 cells/mL of cells wereseeded in a 96-well plate and incubated for 24 hoursat 37ºC. Cells were then treated with the extracts andincubated for 24, 48 or 72 hours. After incubation,20 µL MTT solution (4 mg/mL) was added in eachwell and incubated in 37 C with 5% CO2 saturationfor 4 hours. After the incubation, the medium wasremoved, and the insoluble formazan product wasdissolved in 100 µL of dimethyl sulfoxide.71Absorbance was measured at 570 nm using amicroplate reader. MTT assay was repeated on 48hours and 72 hours. Percentage of inhibition wascalculated as,Inhibition Percentage (%) 100 – ((A570 of treatedcells / A570 of control cells) 100)The value of 50% inhibition (IC50) was calculatedfrom graph which shows the inhibition percentageof viable cells against extract concentrations. Testswere carried out in triplicate.Tail regeneration assayZebrafish for this study has been obtained fromProf. Alexander Chong Shu-Chien, Universiti SainsMalaysia. In vivo activity of various crude extractsfrom B. qriseipurpureus on STZ induced zebrafishwas conducted according to Robert et al. (2013). Allprocedures are performed following the guidelinesdescribed in “Principles of Laboratory Animal Care”(National Institutes of Health publication no. 85-23,revised 1985) and the approved Rosalind FranklinUniversity Institutional Animal Care and UseCommittee animal protocol 08-19.A total of 105 zebrafish was divided into twomain groups which are normal (without STZtreatment; 4 groups of 5 fish each) and induced STZ(STZ treatment; 5 groups of 5 fish each). The STZinduced group were injected with 0.35 mg/g of0.3% streptozotocin on day 1, 3, 5, 12 and 19. Atthis point the zebrafish are considered to have beenin a prolonged state of hyperglycemia and exhibitthe diabetic complications (Robert et al., 2013).0.35 mg/g of 0.09% sodium chloride was used as amock treatment. On day 21, the caudal fin of all fishwas amputated in a straight line proximal to the firstlepidotrichia branching point. Routine examinationon the regenerative growth of caudal fin was doneat 24 and 72 hour following transection. On day 24(72 hours after transection), 5 µL test extracts wereforce feed to the fish for 14 days once daily andcaudal fin regenerative growth was checked andrecorded on day 1, 3, 5, 7 and 14. Metformin (30mg/mL) was used as a positive control drug.Caudal fin regeneration growth rate wascalculated as the percentage of differences of finareas (mm2) grow over the number of days.RESULTS AND DISCUSSIONMycochemical in B. qriseipurpureus extractsThe active ingredient screen of the differentmushroom extracts methods revealed the presenceof alkaloids, flavonoids, saponins and tanins in allavailable forms (hot water, cold water and methanol
72STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusantiviral and antifungal activity and causeshypoglycemia (Francis et al., 2002). In traditionalmedicine, especially in Japan and China, plantextracts containing tannins has been used to treatdiarrhea, abdominal pain and duodenal tumor(Bruyne et al., 1999). Therefore, the presentcompounds in B. qriseipurpureus extracts indicatethe medicinal properties of this species that need tobe discovered.extract). Reducing sugars and anthraquinoneshowever are only present in aqueous extracts (hotwater and cold water) as shown in Table 1. Thepresence of alkaloids, saponins and tannins inextracts is an indication that this species haspharmacological importance (Adebayo et al.,2012). According to Wink et al. (1998) alkaloidshave medicinal properties such as antihypertensitivity (indole alkaloid), antimalaria (quinine),antiarrhythmia (quinidine) and anticancer(vincristine). Anthraquinones and its derivativeshave antitumor properties as it can inhibit celldivision, induce apoptosis and inhibit metastases(Huang et al., 2007). In addition, anthraquinonescan also act as antifungal agent. According to Yaoet al. (2004), most flavonoids have been shown tohave antioxidant activity, free radical scavengingactivity and anticancer activity. Flavanoids alsocapable of preventing coronary heart disease andsome flavonoids demonstrates the ability ofantivirus HIV (Human Immunodeficiency Virus).Extensive research has been done and proven thatsaponins can stimulate the immune system, reducecholesterol levels, has anticancer, antioxidant,Antioxidant properties of B. qriseipurpureusextractsFig. 2 shows the scavenging effect of DPPHagainst different concentrations of the extracts fromB. qriseipurpureus. Hot water extract, cold waterextracts and methanol extract each recorded DPPHscavenging effect of 82.28 0.03, 75.55 0.01 and60.67 0.09 respectively at 10 mg/mL. The DPPHscavenging effect of the control antioxidantcompound, ascorbic acid in this experiment is95.04 0.02%. Our results show that there is acorrelation between the concentrations of theextracts with DPPH scavenging effect. Value ofDPPH scavenging effect increased in parallel withTable 1. Active compounds of the crude extracts from B. qriseipurpureusPhytochemicalsALANFLRSSASTTAHot Water Extract – Cold Water Extract – Methanol Extract – – – , present; –, absent.AL Alkaloids, AN Anthraquinones, FL Flavanoids, RS Reducing Sugars, SA Saponins, ST Steroids,TA TanninsFig. 1. Boletus qriseipurpureus.A: Top View. B: Side View.
STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureus73Fig. 2. DPPH scavenging effect of the crude extracts from B. qriseipurpureus.increasing extract concentration, suggesting that allof the extracts in this study have antioxidantactivity.IC50 is the minimum concentration of substraterequired to inhibit 50% of DPPH free radicals. Bydetermining the IC50 value, a sample can be saidto have either high or low antioxidant activity.The concentration of the extracts from B.qriseipurpureus required to inhibit 50% of DPPHis shown in Table 2. Hot water, cold water andmethanol extract from B. qriseipurpureus eachrecorded IC50 of 1.79 mg/mL, 1.97 mg/mL, and 3.98mg/mL respectively. Overall, the hot water extractand cold water extract showed good antioxidantpotency compared to the methanol extract of B.qriseipurpureus.Previous studies on fruiting bodies of B. edulisextracts showed a lower IC50 values for hot waterextract (0.57 mg/mL) followed by ethanol extract(0.62 mg/mL), cold water extract (0.66 mg/mL)and methanol extract (0.73 mg/mL) (Emanuel &Sultana 2013). This shows that extracts of B. edulismushrooms have better antioxidant potency thanB. qriseipurpureus.Antiproliferative activity of B. qriseipurpureusextracts treated cell linesIn vitro antiproliferative assay of hot water, coldwater and methanol extracts from B. qriseipurpureushad shown that growth inhibition correlates withincreasing concentration of B. qriseipurpureusextracts. Fig. 3 and Fig. 4 showed the growthTable 2. IC 50 of DPPH scavenging effect by the crudeextracts from B. qriseipurpureusTest sampleIC50 of DPPH scavenging effect(mg/mL)Ascorbic Acid0.34Hot Water Extract1.79Cold Water Extract1.97Methanol Extract3.98reduction of breast cancer cell line (MCF-7) andVero cell line against different concentrations ofB. qriseipurpureus extracts at 72 hours after treating.The figs. show that inhibition percentage of MCF-7cell line is higher compared to Vero cell line. Forexample, at the concentration of 100 mg/mL, hotwater extract from B. qriseipurpureus showedinhibition percentage of 60.45% 0.02 and96.42% 0.04 on Vero and MCF-7 cell linesrespectively. This proves the antiproliferative natureof the extract against MCF-7 cell line but theinhibition takes place after 72 hours of treatment.IC50 in antiproliferative assay is the minimumconcentration of substrate required to inhibit 50%of cell viability. By determining the IC50 value, asample can be said to have either high or low antiproliferative activity. The concentrations of theextracts from B. qriseipurpureus required to inhibit50% Vero cells and MCF-7 cells viability are shownin Table 3 and Table 4 respectively. Hot water
74STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusFig. 3. Inhibition percentage of the crude extracts from B. qriseipurpureus on MCF-7 cell line (A) and Vero cellline (B) after 72 hours.extract from B. qriseipurpureus recorded thelowest antiproliferative activity on Vero cell line(IC50, 65.63 mg/mL) and highest antiproliferativeactivity on MCF-7 cell line (IC 50 , 9.79 mg/mL)compared to all extracts tested.A recent study that was conducted using sixspecies of Boletus (B. aereus, B. reticulatus, B.purpureus, B. rhodoxanthus, B. atanas and wildBoletus species) shows that these species containmetabolite compounds especially secondarymetabolites such as phenolic acids (Sandrina et al.,2011) which provides the ability for antioxidant andanticancer activity. Through the antiproliferative testit is assumed that B. qriseipurpureus also containsphenolic acids that leads to anticancer activitytowards MCF-7 cell line.Regeneration of B. qriseipurpureus extractstreated STZ induced fish tailAccording to Robert et al. (2013), the diabeticzebrafish shows decrease in body weight anddelayed caudal fin regeneration growth. Observationby weight of zebrafish taken in this study is alsosimilar to that recorded by Robert et al. (2013).Table 5 and Table 6 shows the weight of each groupof normal and STZ induced zebrafish during theinduction of diabetic (normal group was injectedwith sodium chloride) and after treatment of 14 days.
STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusFig. 4. Percentage of caudal fin regeneration growth rate of the normal fish feed with hot water extract (A), coldwater extract (B) and methanol extract (C) from B. qriseipurpureus after transection.Percentage growth rate of the caudal fin regeneration were calculated as, ((Area of new grown fin (mm 2 ) – Areaof new grown fin on day 3 (mm 2 ))/Total of area of new grown fin (mm 2)*100)/ Number of days. (p 0.05).75
76STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusFig. 5. Percentage of caudal fin regeneration growth rate of the STZ induced fish feed with hot water extract (A),cold water extract (B) and methanol extract (C) from B. qriseipurpureus after transection.Percentage growth rate of the caudal fin regeneration were calculated as, ((Area of new grown fin (mm 2 ) – Areaof new grown fin on day 3 (mm 2 ))/Total of area of new grown fin (mm 2)*100)/ Number of days. (p 0.05).
77STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusFig. 6. Image of regenarative caudal fin growth from STZ induced zebrafish after 14 days post-transection. A.No treatment; B. With treatment of 135 mg/mL hot water extract from Boletus qriseipurpureus; C. With controltreatment of 30 mg/mL metformin.The red dotted line represents the amputation plane and RG demarks the regenerative growth.Table 3. IC50 of Inhibition on Vero cell line by the crude extracts from B. qriseipurpureusTest sampleIC50 on 24 HoursIC50 on 48 HoursIC50 on 72 Hours––––96.7 µg/mL–––35.0 µg/mL65.63 mg/mL48.04 mg/mL43.62 mg/mLTamoxifenHot Water ExtractCold Water ExtractMethanol ExtractTable 4. IC50 of Inhibition on MCF-7 cell line by the crude extracts from B. qriseipurpureusTest sampleTamoxifenHot Water ExtractCold Water ExtractMethanol ExtractIC50 on 24 HoursIC50 on 48 HoursIC50 on 72 Hours88.2 µg/mL–––61.6 µg/mL–––24.6 µg/mL9.79 mg/mL11.48 mg/mL16.85 mg/mLTable 5. Weight of normal fish during the injection (with sodium chloride) phase and after treatment for 14 days. Eachvalue represents the mean value SD of n 5 fish in each groupWeight of Fish (g)GroupWithout TreatmentHWE 45 mg/mLHWE 90 mg/mLHWE 135 mg/mLCWE 45 mg/mLCWE 90 mg/mLCWE 135 mg/mLME 45 mg/mLME 90 mg/mLME 135 mg/mLI1I2I3I4I5T 140.354 0.0870.354 0.1230.355 0.0960.358 0.0640.352 0.0710.356 0.0810.358 0.1010.354 0.0420.352 0.0340.352 0.0530.355 0.0650.354 0.0540.356 0.0650.359 0.0860.353 0.0450.357 0.0890.360 0.0840.355 0.0820.353 0.0730.352 0.0540.356 0.0780.356 0.0830.357 0.0930.361 0.0910.353 0.1020.357 0.0830.361 0.1150.356 0.0760.354 0.0930.353 0.1340.362 0.0980.362 0.1020.362 0.8900.363 0.1230.358 0.1420.362 0.0780.364 0.0880.364 0.0980.361 0.0650.359 0.0760.367 0.0780.366 0.0870.367 0.0980.368 0.1080.364 0.0870.367 0.7600.370 0.0910.369 0.1030.365 0.0540.364 0.0650.377 0.1430.377 0.0920.375 0.0830.371 0.0730.372 0.0630.375 0.0830.381 0.0520.376 0.1390.373 0.0620.376 0.870HWE Hot Water Extract, CWE Cold Water Extract, ME Methanol Extract, I Injection, T 14 Treatment Day 14.
78STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusTable 6. Weight of STZ induced fish during the diabetic induction phase and after treatment for 14 days. Each valuerepresents the mean value SD of n 5 fish in each groupWeight of Fish (g)GroupWithout TreatmentHWE 45 mg/mLHWE 90 mg/mLHWE 135 mg/mLCWE 45 mg/mLCWE 90 mg/mLCWE 135 mg/mLME 45 mg/mLME 90 mg/mLME 135 mg/mLMetformin 30 mg/mLI1I2I3I3I4T 140.356 0.0580.355 0.0620.353 0.0720.356 0.0560.354 0.0530.351 0.0650.357 0.0780.353 0.0540.358 0.0650.359 0.0760.354 0.0940.356 0.0870.355 0.0780.353 0.0360.355 0.0870.354 0.0710.351 0.0540.356 0.0760.353 0.0390.358 0.0870.358 0.0540.353 0.0760.355 0.0980.353 0.1320.352 0.0920.353 0.0450.353 0.0430.349 0.0980.355 0.0670.351 0.0340.356 0.0650.356 0.0670.352 0.0730.353 0.0880.348 0.0790.347 0.0870.349 0.0650.348 0.0760.344 0.0490.352 0.0670.346 0.0870.350 0.0870.350 0.0850.348 0.0760.347 0.0890.343 0.0870.341 0.1030.342 0.0890.342 0.1240.339 0.0780.347 0.1320.340 0.0760.344 0.0560.345 0.0890.341 0.1070.344 0.0920.349 0.0890.349 0.1340.352 0.1560.343 0.0780.342 0.0540.350 0.0430.341 0.08870.349 0.01060.351 0.15550.353 0.167HWE Hot Water Extract, CWE Cold Water Extract, ME Methanol Extract, I Injection, T 14 Treatment Day 14.STZ induced zebrafish showed weight loss duringthe phase of diabetic induction, while the normalfish increased in weight. However, increased in bodyweight in STZ induced group was seen after thetreatment with the B. qriseipurpureus extract.Caudal fin regeneration growth rate of thenormal fish and STZ induced fish feed with crudeextracts from B. qriseipurpureus is shown in Fig. 4.Continuous caudal fin growth was observed in bothgroups. However growth rate was seen to decreaseafter the 6th day (normal fish) and 10th day (STZinduced fish) of post transection. Normal fish feedwith hot water extract recorded higher caudal fingrowth rate compared to fish feed with otherextracts. Among the hot water extract, the highestconcentration (135 mg/mL) showed the highestcaudal fin growth rate in normal fish. Similartrend was seen in STZ induced fish fish; fish feedwith 135 mg/mL hot water extract showed highestcaudal fin growth rate comparable to the fish fedwith the positive control drug (metformin). Theresults suggest that hot water extract from B.qriseipurpureus may cause hypoglycemic withinthe fish and the activity increases with the increasein the extracts concentration.CONCLUSIONOur findings show that, the hot water extract of B.qriseipurpureus has the maximum antiproliferativeactivity, DPPH scavenging activity and antidiabeticactivity. Antioxidant activity and cytotoxicity effecthas positive linear relationship (Wing et al., 2007)and could delay the development of complicationsin diabetic mellitus (Fatimah et al., 2012). Thisevidence supports the oral statements of the localpeople that claimed gelam mushroom could be usedas a remedy for diabetes. The present study wasrestricted to the preliminary screening of activecompounds, antioxidation, antiproliferative andantidiabetic test of the selected extracts. Furthercharacterization and identification of compoundswithin the hot water extract of B. qriseipurpureusneed to be done before this extract can be used asan alternative remedies to defeat cancer anddiabetes.ACKNOWLEDGEMENTSThis project was financially supported by the grantFRGS/1/2013/ST03/UKM/02/4, GGPM-2014-006and FRGS/2/2014/ST04/UKM/03/1.REFERENCESAhmad, R., Muniandy, S., Shukri, N.I.A., Alias,S.M.U., Hamid, A.A., Yusoff, W.M.W., Senafi, S.& Daud, F. 2014. Antioxidant properties andglucan compositions of various crude extractfrom Lentinus Squarrosulus mycelial culture.Advances in Bioscience and Biotechnology 5:805-814.Adebayo, E., Oloke, J., Ayandele, A. & Adegunlola,C. 2012. Phytochemical, antioxidant andantimicrobial assay of mushroom metabolitefrom Pleurotus pulmonarius. Journal ofMicrobiology and Biotechnology Research 2:366-374.
STZ INDUCED ZEBRAFISH OF VARIOUS CRUDE EXTRACTS FROM Boletus qriseipurpureusAyoola, G., Coker, H., Adesegun, S., Adeoju-Bello,A., Obaweya, K., Ezennia, E. & Atangbayila, T.2008. Phytochemical screening and antioxidantactivities of some selected medicinal plants usedfor Malaria therapy in Southwestern Nigeria.Tropical Journal of Pharmaceutical Research7: 1019-1024.Barros, L., Baptista, P., Estevinho, L.M. & Ferreira,I.C.F.R. 2007a. Effect of fruiting body maturitystage on chemical composition and antimicrobial activity of Lactarius sp. Mushrooms.Journal of Agricultural and Food Chemistry55: 8766-8771.Barros, L., Calhelha, R.C., Vaz, J.A., Ferreira,I.C.F.R., Baptista, P. & Estevinho, L.M.2007b. Antimicrobial activity and bioactivecompounds of Portuguese wild ediblemushrooms methanolic extracts. EuropeanFood Research and Technology 225: 151-156.Bruyne, T.D., Pieters, L., Deelstra, H. & Vlietinck,A. 1999. Condensed vegetable tannins:Biodiversity in structure and biologicalactivities. Biochemical Systematics andEcology 27: 445-459.Chen, J.N., Wang, Y.T. & Wu, J.S.B. 2009. Aglycoprotein extracted from golden oystermushroom pleurotus citrinopileatus exhibitinggrowth inhibitory effect against U937 leukemiacells. Journal of Agricultural and FoodChemistry 57: 6706-6711.Dore, C., Azevedo, T.C.G., de Souza, M.C.R., Rego,L.A., de Dantas, J.C.M. & Silva, F.R.F. 2007.Antiinflammatory, antioxidant and cytotoxicactions of betaglucan rich extract from Geastrumsaecatum mushroom. International Immunopharmacology 7: 1160-1169.Emanuel, V. & Sultana, N. 2013. Antioxidantcapacity and the correlation with majorphenolic compounds, anthocyanin, andtocopherol content in various extracts fromthe wild edible Boletus Edulis mushroom.BioMed Research International 1-11.Faccin, L.C., Benati, F., Rincao, V.P., Mantovani,M.S., Soares, S.A. & Gonzaga, M.L. 2007.Antiviral activity of aqueous and ethanolextracts and of an isolated polysaccharide fromAgaricus brasiliensis against poliovirus type 1.Letters in Applied Microbiology 45: 24-28.Fang, F. 2013. Phylogenetic analysis of the Asiancyprinid genus Danio (Teleostei, Cyprinidae).Copeia 4: 714-728.Fatimah, A.M., Siti, B.B., Zariyantey, A.H.,Nasar, A. & Jamaludin, M. 2012. The role ofoxidative stress and antioxidants in diabeticcomplications. Sultan Qaboos UniversityMedical Journal 12: 1(5-18).79Francis, G., Kerem, Z., Makkar, H.P. &
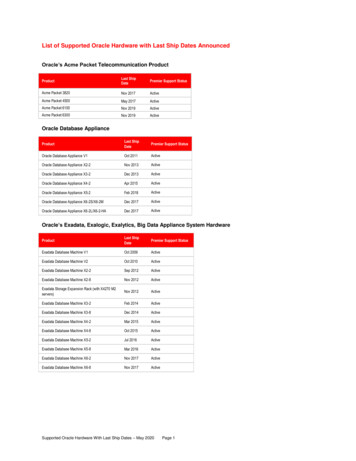
Scavenging effect (%) (A Blank - A Sample / A Blank) 100 Where A Blank is the absorbance of the control reaction (containing all reagents except the test compound), and A Sample is the absorbance of the test compound. The value of 50% inhibition (IC 50) was calculated from the graph plotted inhibition percentage against extract .